WHO issues guidance on GLP-1 drugs for obesity, calls them a "scientific breakthrough" and "new chapter."
The World Health Organization (WHO) marked a pivotal moment in global public health on Monday, December 1, 2025, by releasing comprehensive new guidelines endorsing the use of GLP-1 receptor agonist drugs for the treatment of obesity in adults. This landmark guidance, published and widely reported, including by CBS News, hails these medications not merely as a "scientific breakthrough" but as inaugurating a "new chapter" in the global understanding and management of obesity. The announcement signals a significant conceptual shift, moving obesity from a frequently stigmatized "lifestyle condition" to a recognized complex, preventable, and treatable chronic disease requiring comprehensive, lifelong care.
At the core of the WHO’s new directive are two key recommendations. Firstly, the agency advises that GLP-1 therapies may be utilized by adults for the long-term management of obesity. A crucial caveat, however, excludes pregnant women from this recommendation, underscoring the need for careful patient selection and medical supervision. Secondly, the guidelines strongly advocate for the concurrent application of intensive behavioral therapy, encompassing significant adjustments related to diet and regular physical exercise, in conjunction with the pharmacological treatment. This dual approach emphasizes that medication is a powerful tool, but not a standalone solution, integrating seamlessly with established lifestyle interventions.

Dr. Tedros Adhanom Ghebreyesus, the Director-General of the WHO, articulated the profound implications of this guidance in a news release. He stated, "Obesity is a major global health challenge that WHO is committed to addressing by supporting countries and people worldwide to control it, effectively and equitably. Our new guidance recognizes that obesity is a chronic disease that can be treated with comprehensive and lifelong care." Dr. Ghebreyesus further acknowledged the transformative potential of these drugs, adding, "While medication alone won’t solve this global health crisis, GLP-1 therapies can help millions overcome obesity and reduce its associated harms." His words underscore a measured optimism, balancing the revolutionary impact of GLP-1s with the systemic challenges inherent in addressing a widespread public health crisis.
Despite the enthusiastic reception and the recognition of their immense potential, both recommendations from the WHO are designated as "conditional." This classification reflects a cautious yet progressive stance, acknowledging areas where further research and implementation strategies are required. The recommendation for GLP-1 use in adults is deemed "conditional" primarily due to several factors: limited long-term data regarding the medications’ efficacy and safety over decades, the substantial costs associated with these treatments, and other practical considerations such as the need for sustained medical oversight and potential side effects. Similarly, the recommendation for intensive behavioral therapy alongside GLP-1s is also graded "conditional," based on "low-certainty evidence" specifically concerning whether this combined approach demonstrably enhances GLP-1 treatment outcomes beyond what the medication achieves on its own. This does not diminish the importance of lifestyle changes, but rather highlights a need for more robust studies on their synergistic effect with GLP-1s.
The narrative of GLP-1 drugs as a "new chapter" is further elaborated in an article published concurrently in the peer-reviewed medical journal JAMA. Co-authored by Francesca Celletti, a senior advisor for obesity at WHO’s Nutrition and Food Safety department, the article posits, "They represent a new chapter in the gradual conceptual shift in how society approaches obesity – from a ‘lifestyle condition’ to a complex, preventable, and treatable chronic disease." This shift is monumental, moving away from a historically simplistic and often judgmental view of obesity as a failure of willpower, towards a nuanced medical understanding. For decades, obesity was largely seen as a personal failing, a result of poor dietary choices and lack of exercise, leading to widespread stigma and inadequate medical intervention. The WHO’s official recognition fundamentally redefines this perception, paving the way for more compassionate care, increased research funding, and improved access to effective treatments.
The global burden of obesity is staggering, affecting more than 1 billion people worldwide and contributing to an estimated 3.7 million deaths last year alone, according to WHO figures. In light of this immense challenge, the organization highlighted the critical importance of equitable access to GLP-1 drugs. It issued a fervent call for "urgent action on manufacturing, affordability, and system readiness to meet global needs." This plea acknowledges the significant disparities in healthcare access and economic resources across different nations. The current landscape suggests a grim reality: even with a projected rapid expansion in production capacity, GLP-1 therapies are estimated to reach fewer than 10% of individuals who could potentially benefit from them by the year 2030. This projection underscores a looming global health equity crisis, where the benefits of a scientific breakthrough might remain inaccessible to the majority of those who need it most, particularly in low- and middle-income countries. Addressing this will require concerted efforts from governments, pharmaceutical companies, and international health organizations to ensure sustainable production, fair pricing, and robust distribution networks.
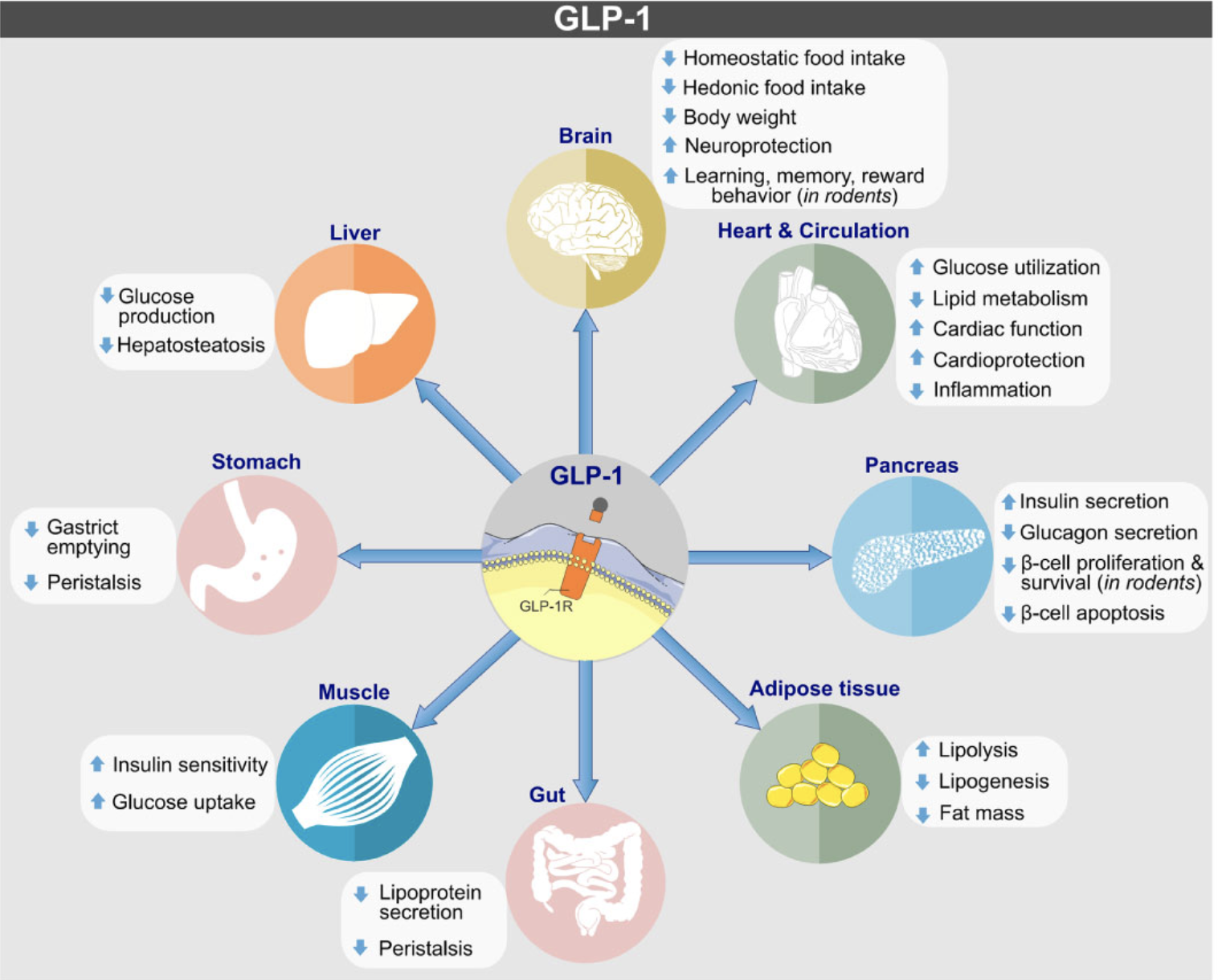
GLP-1 receptor agonists, such as semaglutide and tirzepatide, function by mimicking the natural hormone glucagon-like peptide-1, which is released in the gut in response to food intake. These drugs activate GLP-1 receptors, leading to several physiological effects beneficial for weight management and blood glucose control. They stimulate insulin secretion, suppress glucagon secretion, slow gastric emptying, and crucially, act on appetite centers in the brain to reduce hunger and increase feelings of fullness. While highly effective, common side effects can include nausea, vomiting, diarrhea, and constipation, usually more pronounced at the beginning of treatment and often dose-dependent. More serious, though rare, side effects can also occur, necessitating careful medical supervision.
In the United States, several well-known brand names exemplify the GLP-1 drug class. Ozempic and Mounjaro are widely recognized and FDA-approved primarily for treating type 2 diabetes. Their weight-loss effects were initially observed as a beneficial side effect. Subsequently, higher-dose formulations, such as Wegovy (semaglutide) and Zepbound (tirzepatide), received specific FDA approval for chronic weight management in individuals with obesity or overweight with at least one weight-related comorbidity. The WHO’s inclusion of GLP-1 therapies on its "Essential Medicines List" in September for managing type 2 diabetes in high-risk groups served as a precursor to this broader obesity guidance, signaling a growing recognition of their fundamental role in modern medicine.
The WHO’s guideline represents a bold step forward in addressing the global obesity epidemic. By endorsing GLP-1 therapies within a framework of comprehensive care, the organization aims to empower individuals living with obesity and support healthcare systems in providing effective treatment. This comprehensive approach, as reiterated by the agency, must encompass not only medication but also healthy diets, regular physical activity, and sustained support from qualified health professionals. The challenges are immense, ranging from ensuring equitable access and managing costs to conducting further long-term research on efficacy and safety, particularly in diverse populations. Nevertheless, the declaration of GLP-1 drugs as a "scientific breakthrough" and the opening of a "new chapter" offers a beacon of hope for millions struggling with this chronic disease, fundamentally reshaping the global health discourse on obesity.